侧边栏
Preparation of covalently cross-linked sulfonated polybenzimidazole membranes for vanadium redox flow battery applications
J. Membr. Sci. 2017, 525, Pages 229–239 (1 March 2017)
By Zijun Xia, Libin Ying, Jianhua Fang*, Yu-Yu Du, Wei-Ming Zhang*, Xiaoxia Guo, Jie Yin
j.memsci.2016.10.050, local fulltext
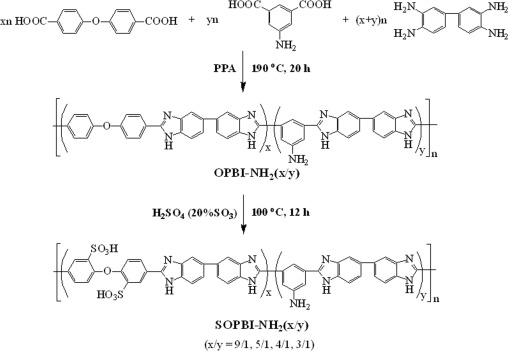
A series of polybenzimidazole copolymers with varied content of pendant amino groups have been synthesized by condensation polymerization of 4,4′-dicarboxydiphenyl ether (DCDPE), 5-aminoisophthalic acid (APTA) and 3,3′-diaminobenzidine (DAB) in polyphosphoric acid at 190 °C for 20 h. The resulting copolymers undergo post-sulfonatation in fuming sulfuric acid at 100 °C yielded the highly sulfonated polybenzimidazoles (SOPBI-NH2(x/y), ‘x/y’ refers to the monomer molar ratio of DCDPE to APTA). A series of covalently cross-linked membranes (CSOPBI-NH2(x/y)) with good mechanical properties are fabricated by solution cast technique using bisphenol A epoxy resin as a cross-linker. The CSOPBI membranes show 3–4 orders of magnitude lower VO2+ permeability and 6–30 times higher ion diffusion selectivity (proton vs. VO2+) than Nafion117. The charge-discharge behaviors of the vanadium redox flow batteries (VRBs) assembled with the CSOPBI-NH2(x/y) membranes and Nafion 117 are investigated and compared. The VRBs assembled with the CSOPBI membranes exhibit significantly higher columbic efficiency and lower self-discharge rate than that assembled with Nafion 117 owing to the extremely lower vanadium cations crossover of the former. The VRB assembled with the CSOPBI-NH2(9/1) membrane exhibits fairly high energy efficiency (~85% at 60 mA cm−2) and little decay in performance is observed after 300 charge-discharge cycles.